In 2013, the launch of White Lyan in London put sustainability into the hospitality world in a big way. It was a bar with no ice or fruit, an idea which challenged the way bartenders thought about creating drinks. With no citrus to lean on, the White Lyan team developed a number of acid-rich solutions to balance their drinks.
Since then, bartenders across the globe have been dropping acid in drinks, and experimenting with acidity as a way to tweak or manipulate other ingredient liquids; this is known as ‘acid adjusting’ or acidulating.
Yet although these can be impressive substitutes, these kind of acid solutions are still in many ways an imperfect solution. Personal experience has taught me the acidity can often play havoc with your own palate, and give some guests undesirable throat burn as they punish their third acid-laden cocktail.
As noted by Liquid Intelligence author, Dave Arnold, the acidulated liquid can often be ‘relatively thin and missing any viscosity.’
When you taste an acidulated solution against the real thing, it’s clear that there is an element of the acidulated solution that tastes fake, and without the use of other flavours to back up the acidity it is difficult to create a well rounded flavour. Acid adjusting works to create balance, yes, but you also need to ensure the complexity of the final drink.
While the use of acid solutions are nothing new, a few forward-thinking bars have found that using them to amplify existing flavours in ingredients is an alternate way in which bartenders can balance a drink without the use of citrus. It pays to keep in mind that by understanding these different types of acids, you can manipulate texture and enhance flavour.
Before we take a deep dive into the adjustment of acidity in liquid, we need to first define it, and look at how they present themselves differently in each form. By adjusting acidity, we are playing with the pH (potential of hydrogen) of an aqueous liquid (liquid containing water) which refers to how acidic or alkaline it is based on its hydrogen ion concentration. The pH scale is a numeric scale, running from 0 to 14 and is broken down like this:
- A pH of 0 indicates high level of acidity
- A pH of 7 is neutral
- A pH of 14 indicates it is alkaline
Food that is considered acidic must have a pH level of 4.6 or lower, so when looking at lemon (approx. 2.00-2.60 and with 0.6g sugar per 30ml liquid) and lime (approx. 2.00-2.80 and with 0.2g sugar per 30ml liquid) they are both on the heavily acidic scale. When looking at an orange with a pH of between 3.69-4.34 and with 3.1g sugar per 30ml liquid, you can start to look at how the use of citric or malic acid can manipulate the pH of orange juice to create a liquid reminiscent of lemons or limes in a cocktail but with the texture and base flavour notes of orange.
One who knows the technique better than most is Zach Morgan (ex-Bulletin Place, currently at Black Pearl).
“Juices such as pineapple, orange, or grapefruit can be seasoned with powdered citric, malic, tartaric, lactic or a specific blend of the former to maintain a more consistent product throughout the year,” he says, as fruits vary in pH levels depending on ripeness, time of the season they were harvested and the origin of the fruit. Classic cocktails that require fresh juice are most affected by this, with a Blood and Sand or Mary Pickford two examples Morgan mentions that can be drastically improved with the acid adjustment.
Through this technique, you can acid-adjust grapefruit juice in a drink to transform the juice into a brighter, more cocktail-friendly ingredient; for example, acid-adjusting grapefruit juice in a Daiquiri riff, or replacing the lemon juice with acid-adjusted orange juice in a Sidecar or a Whisky Sour. Or maybe you’d like to acid-adjust an oleo-saccharum and create a complex, interesting cordial.
Whatever the application, taking the time to measure the pH of a liquid and experinmenting with acid-adjusting can give your customers a better drinking experience, and open up new creative avenues.
Drop these acids (in your drinks)
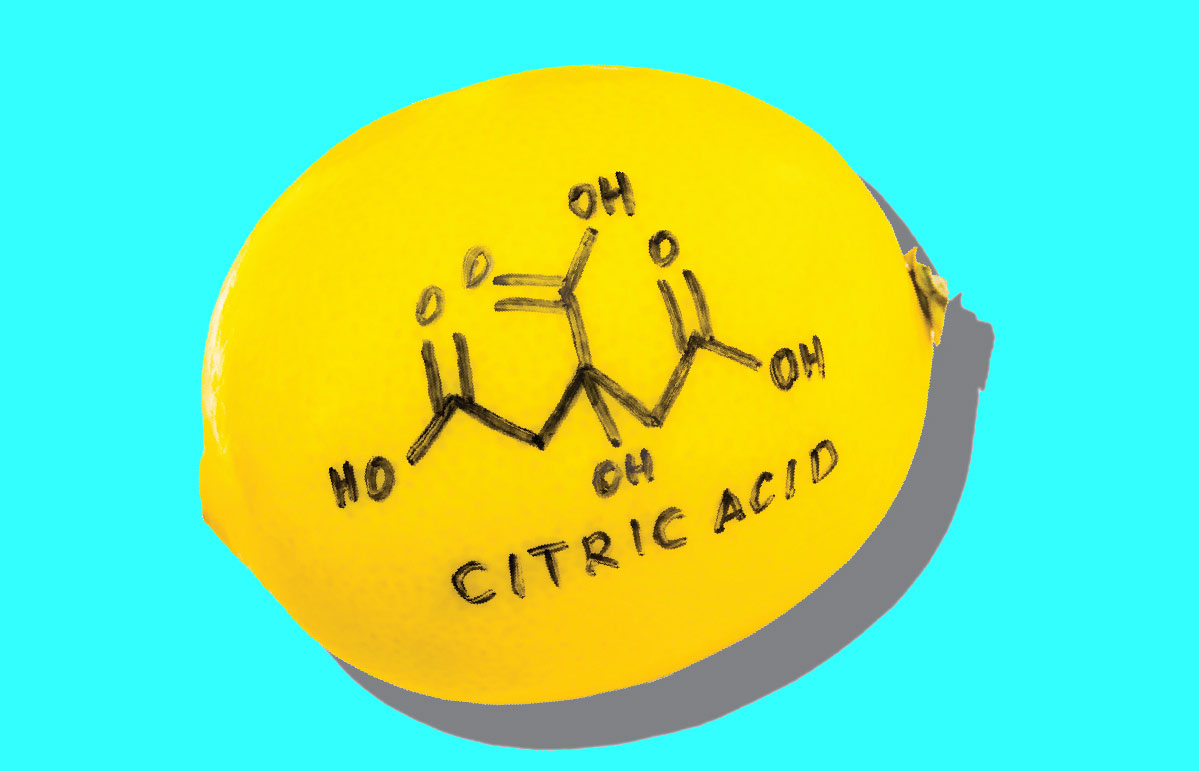
Citric Acid
An organic acid, citric acid is found in both lemons and limes in high quantities, can be described as acerbic when overused, and is closely associated with lemons.
Malic Acid
Also an organic acid, malic acid is not as harsh as citric acid, it is closer to biting into a juicy granny smith apple, grapes or rhubarb; it’s refreshing and mouth-puckering.
Lactic Acid
Is an organic acid and what gives yoghurt or kefir a tang, and can enhance savoury flavours or round off the edges of a drink; it adds a fuller, creamy mouthfeel to cocktails, and has a slightly higher pH that citric or malic acids.
Lyan uses it in a creamy Martini to give a creaminess to the palate.
Phosphoric acid (inorganic)
As this doesn’t occur naturally — it is inorganic — we don’t link it with anything we’ve tasted before. It does not impart any additional flavour, but gives existing flavours a boost. Iit can highlight a delicate pineapple acidity in a drink and give soda an extra spark. It can be used to turn solid minerals into tinctures: White Lyan used it in a Bone Dry Martini, where the acid dissolves a chicken bone into a tincture creating an acid, mineral and dry finish. Use just a drop or two of phosphoric acid solution, made at a 1.25% phosphoric acid to solution ratio.
Tips & Tricks:
- Acids are by nature corrosive and should always be treated with care when included in drinks.
- Always make sure they are food-grade.
- Keep acidic solutions in glass containers as to ensure they don’t react with metal or plastic.
- Acid adjustment of liquids should be treated similar to seasoning in cooking, it’s a way to elevate existing flavours.
















